Sodium emission spectrum9/16/2023  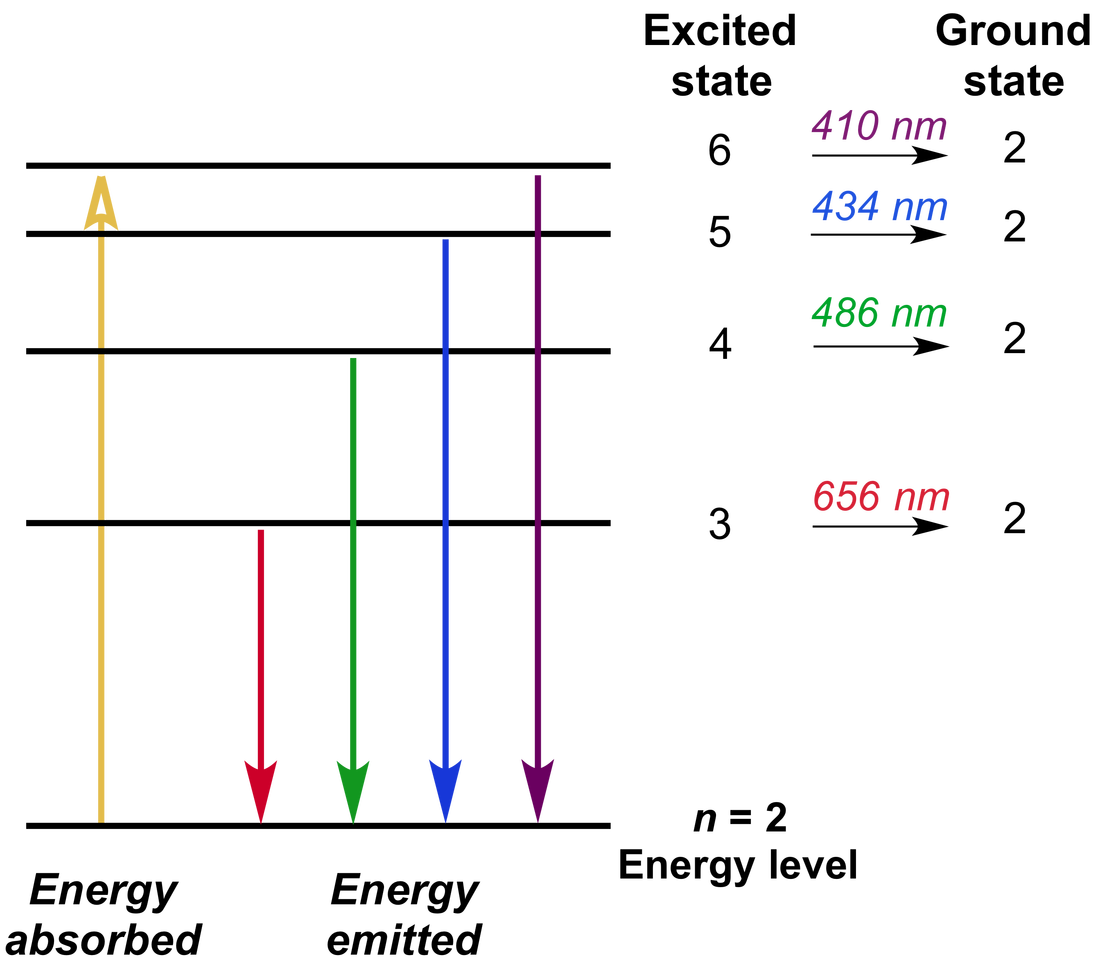
Light from distant stars and galaxies act like the incandescent bulb in our previous example: this light has certain absorption lines from its travels, which can be used to determine what gases are in the intergalactic and interstellar media and in what quantities. Interestingly, astronomers use exactly this technique to study gases in the vacuum of space, in between stars in our galaxy and in between galaxies in our universe. The compilation includes data for the neutral and singly-ionized atoms of all elements hydrogen through einsteinium ( Z 1-99). This would cause an absorption spectrum of sodium in the light you observe from the incandescent bulb. Sodium Green (sodium bound) is a fluorescent compound with an excitation peak at 507 nm and an emission peak at 532 nm. Introduction This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily accessible format. The sodium in between you and the lamp absorbs photons at certain wavelengths (the same ones it was emitting at earlier!) while the electrons in the sodium are excited to higher energy levels. The incandescent bulb emits visible light at all wavelengths. The answer to your second question is yes! Imagine there is some background source, like a regular incandescent light bulb, behind your sodium lamp. Find Emission Spectrum Sodium Element stock images in HD and millions of other royalty-free stock photos, illustrations and vectors in the Shutterstock. For this reason, low-pressure sodium lamps are used. Then, instead of seeing the sharp spectral lines that you are used to seeing, you would see blurred lines. The energy of an emitted photon is exactly equal to this change of energy, so instead of having one energy at which many photons are emitted, you have a range of energies through which these photons are emitted. That changes the energies that the different states could have just a little bit. When there are two atoms very close to each other, they interact, via the electric force. Lines in the AtomTrace list were observed in the optical emission of laser induced plasmas, and the respective values of spectroscopic constants are taken from 1. These patterns are based on the physics of one atom by itself. The three atomic emission spectra for Sodium can be shown on graph bellow: NIST Atomic Spectra Database 1, gfall.dat file from Kurucz CD-ROM No. Since they only occur at certain energies (and frequencies and wavelengths), we observe a spectral line pattern. When they do, they emit a photon at the energy equal to the change of energy of the electron these photons are what we observe. Therefore, these excited sodium atoms drop down to lower energy states very quickly. These states exist at higher energies than the ground states, but are unstable. In a sodium lamp, electrons excite sodium atoms from the ground state to excited states. Examining the emission and absorbtion lines of chemical elements. You have the right idea of how these spectral lamps work. Visible light spectral emission lines from sodium in a flame. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
